Fichas sobre get, creado por yousef mohamed el 10/06/2019.
Pineado a
5
1
0
Sin etiquetas
|
|
Creado por yousef mohamed
hace alrededor de 5 años
|
|
Cerrar
|
|
Creado por yousef mohamed
hace alrededor de 5 años
|
|

benzyl.penicillin
phenoxy-methyl penicillin
fused B-lactam + thiazolidine bicyclic structure
6-APA
4-Thia-1-azabicyclo [3,2,0] heptane
6-phenyl.acetamido-2,2-dimethyl-penam-3-carboxylic acid.
(4-Thia-1-azabicyclo13.2.0] hept-2-ene) 7-one
(1-Azabicyclo (3.2.0) hept-2-ene)-7-one
(5-Thla-1-azabicyclo. [4.2.0]oct-2-ene)-8-one
1-Azacyclobutan-4-one
2,6-dimethoxy-benzamido penicillanic acid
5-thia-1-azabicyclo [4, 2,0] octanes
B-lactam + dihydro.thiazine
3,3-dimethyl-7-oxo-4-thia-1-azabicyclo [3.2.0] heptane-2-carboxylate 4,4- dioxide.
(2S,5R,6R)-6-[(R)-2-amino-2-phenylacetamido ] -3,3 dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2 carboxylate.
Have weak activity comparable to 6- APA
fused B-Lactam + oxazolidine ring
contain one ring [azetidine
pKa values between 6.0 and 9.0.
2,3,6-trihydroxy -3- methoxy-3-C-methyl-L-ribo-hexose
6-methyl ether of erythromycin
prototype of a series of nitrogen-containing, 15-membered ring known as azalides.
2,2-Dichroro-N-[(IR,2R)-2-hydroxy-1- hydroxy-methyl-2-(4-nitrophenyl) ethyl acetamide.
1945
1960
late 1970s
1980s
oral bioavailability good, (40%)
Effective concentrations achieved in bone for staphylococcal osteomyelitis but not in cerebrospinal fluid for meningitis
below Pka 10.4
protect against, cure, streptococcal in mice
aniline-substituted
2. Prodrugs react to generate active sulfanilamides
3. Non-aniline sulfonamides
Synthesis:
1. Reaction p-nitrobenzenesulfonyl-chloride + amine the intermediate + reduction leads directly
a. Short-acting sulfonamides:
b. Intermediate-acting sulfonamides:
c-Long-acting sulfonamides:
For: cholera, leprosy and malaria.
4-amino-N- (4,6-dimeth yl-2 - pyrimidinyl) benzene surfonamide.
4-amino-N-(-2-pyrimidinyl) benzene sulfonamide.
4- amino-N- (5, 6-dimethoxy-4pyrimidinyl) benzene sulfonamide.
4-amino-N-(6-methoxypyridazin-3-yl) benzene sulfonamide
Intestinal sulfonamides
2-hydroxy-5- [ [(4- [2pyridinyl.amino) sulfonyl]-phenyl] Azo] benzoic acid.
Urinary sulfonamides
Ophthalmic sulfonamides:
Burn therapy sulfonamides:
4-amino-N-(3,4-dimethylisoxazol-5yl) benzene sulfonamide
N-[(4-aminophenyl) sulfonyl) acetamide
4- (Amino methyl) benzene sulfonamide.
5- [ (3,4,5-trimethoxy phenyl) methyl] pyrimidine-2,4-diamine
sulfamethoxazole- trimethoprim (Co-trimoxazole) (septazole, sutrim), to prevent
N'-alkylated-3-carboxypyrid-4-one ring
(1997-1999).
(1965
I-Ethyl-1,4-dihyro-7-methyl-4-oxo- 1,8-naphthyridine-3-carboxylic
1-Cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinoline-3-carboxylic acid
(+/-) 6-Fluoro-7-piperazinyl quinolone with methyl substituted oxazine ring
acute bacterial sinusitis
chronic bronchitis
tri-fluoro.naphthyridine derivative with aza-bicyclo group at C-7 and formulated as hydrochloride or mesylate salt.
3- Alkyl substitution at position 1 is essential
di-fluoro-phenyl group may cause hepatotoxicity
4- Substitution C-5 with amino group improves both Gram +ve and Gram -ve.
This improved dependent on C-8 and is most active when substituent fluorine,
6- Addition of piperazine group at C-7 provides antipseudomonal activity.
Alkyl substitution of piperazine
improves activity against gram +ve bacteria + increase drug's half-life.
Lipophilic aza-bicyclo modification C-7 increases activity Gram +ve
7- Addition second fluorine group at C-8 increases absorption and longer half-life & increases phototoxicity
Iso-nicotinic Acid Hydrazide, INH).
(+) N, N-bis-(1-hydroxy-2-butyl) ethylene-diamine
pyrazine-carboxamide
2-Ethyl-thio-iso-nicotin-amide
fluoro.deoxy.uridine.monophosphate
8S,9R-6 -Methoxy.cinchonane-9-ol
7-Chloro-4 { [ 4-(diethyl.amino)-1 methyl.butyl ) amino } quinoline.
7-Chloro-4 { { 3- (diethyl.amino.methyl) -4-hydroxy.phenyl] amino } quinolone
8- [ ( 4-Amino-1-methyl-butyl) amino ] -6-methoxy.quinoline.
1-(4-chlorophenyl) -5- isopropyl.biguanide
5- (4-Chlorophenyl) -6-ethyl-2,4 pyrimidine.diamine
2-14-(4,6-diamino-1, 3,5-triazin-2-yl-amino) phenyl-1,3,2-dithia.rsolane-4-methanol
Alpha.-Difluoromethylornithine, OR DFMO'
4-[5-nitrofurfurylidene)amino]-3 methylthiomorpholine1,1-dioxide
N-Benzyl-2-nitroimidazole-1-acetamide
2-(2-Methyl-5-nitroimidazolyl)ethanol
5,7-Di.iodo-8-hydroxy.quinoline
2-furoate ester of : 2,2-dichloro-4'-hydroxy-N-methyl.acetamide
5 -chloro-N-(2-chloro-4-nitrophenyl)-2-hydroxy. benzamide
N.N-diethyl -4-methyl-1-piperazine carboxamide.
S)-2,3,5,6-tetahydro-6-phenyl.imidazo-(2,1-b) thiazole hydrochloride
Mebendazole
2-(4-thiazolyl)-1H-benzimidazole
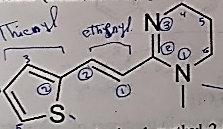
E-1, 4, 5, 6- tetrahydro-1-methyl-2-[2-(2-thienyl) ethenyl-pyrimidine.
2-cyclo.hexyl.carbonyl)-1,2,3,6,7,1.I.b-hexa.hydro-4H-pyrazino[2,1-a] iso.quinolin-4-one.
Tri sod phosphono formate
ethyl (3R,4R,5S)-4-acetamido-5-amino-3-(pentan-3-yloxy)-cyclohex-1-ene-1-carboxylate
Bis (2-chloroethyl)
methylamine HCl
4-[bis(2 chlorethyl) amino] benzenebutanoic acid
2,4,6-Tris(aziridin-1-yl)-1,3,5-triazine
1,4-Bis(methanesulfonyloxy)butane
6-[(1-Methyl-4-nitro-1H-imidazol-5-yl)sulfanyl]-7H-purine
5-Methoxy-2-[(4-methoxy-3,5-dimethylpyridin-2-yl)methanesulfinyl]-1H-benzimidazole
N-Ethyl-N-(2-methylphenyl)but-2-enamide
P-hydroxy benzoic acid derivatives

 Ocultar las fichas que te sabes
Ocultar las fichas que te sabes