16407036
Photosynthetic Metabolism
Description
No tags specified
Flashcards by Alice Hathaway, updated more than 1 year ago
More
Less
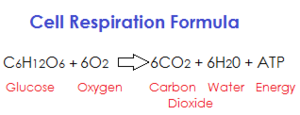
|
Created by Alice Hathaway
over 5 years ago
|
|
Resource summary
| Question | Answer |
| Importance if metabolism | Allows cells to survive and ecosystems to develop Redox reactions drive metabolism, ETC made of mobile carrier and membrane protein complexes Flexible and responds to environment |
| Ways of gaining energy | Photoautotroph Chemoautotroph Heterotroph All have similar core metabolic pathways hence from common ancestor |
| Complex pathways unified by | Biochemical building blocks Energy stores Carrier molecules Regulatory proteins |
| Redox reactions | Split into half reactions Those with more negative potentials donate electrons, accepted by those more positive |
| Yeast fermentation | Sugars from barley Thermodynamically favourable to produce 2ATP Glucose -> pyruvate. Produce 2NADH Won't proceed without ADP or NAD+ |
| Regulation of fermentation | Under anaerobic conditions Gene expression - enzyme expression reduced if on non-fermentable carbon source |
| Why reduced ATP yield of glucose? | Glucose only partially oxidised hence still energy rich Different in reduction potential between primary donor and terminal acceptor relatively small |
| What is external electron acceptor used? | All carbon fully oxidised to release maximal energy To allow electron transfer, requires electron carrier and ETC |
| NADPH and FADH2 | Similar redox carriers FADH2 embedded within proteins - not mobile carrier |
| Electron Transport Chain Structure | Terminal electron donor and acceptor Transmembrane complexes containing electron carriers Mobile carriers to transport electron between complexes Electron or hydrogen |
| Theory of ETC | PMF created to generate ATP by chemiosmosis Mobile electron carriers Mobile hydrogen carriers Large transmembrane protein complexes |
| Mobile electron carriers | Small proteins transition metals to carry electron directly |
| Mobile hydrogen carriers | Organic Carry electron only if undergo chemical reaction involving protons plastaquinone and ubiquinone |
| Large transmembrane protein complexes | Multiple protein subunits Omternal electron acceptors Quinones, haem groups, chlorophyll, FeS clusters Complex II in mitochondria contains haem, 3FeS and FAD |
| Use of ETC regulated and flexible | Chemolithotrophic Bacteria in biomining Mineral ores rich in precious metal in sulphite form insoluble hence expensive to extract Source of Fe3+ act as oxidising agent on sulphite, release as soluble sulfate - extracted by other processes Bacterium provides Fe3+ through ETC |
| Metabolic flexibility in Chemolithotrophic Bacteria | ATP synthesis = Fe2+ oxidised in outer membrane on cytochrome C, forming Fe3+. Periplasmic electron carrier proteins transfer e to cytochrome C oxidase. Transferred to O2 as terminal acceptor NAHD synthesis = ATPase turned off and PMF transport electrons through complexes and carriers in reverse electron flow to produce more NADH Can switch between pathways |
| Challenges for harvesting light | Individual chlorophyll only max 10 photons/ second Water inert, but supplies electrons for ETC Transfer of electrons from water to NADPH needs complex ETC |
| Photosynthetic organisms | Plant, algae, cyanobacteria Green algae - unicellular/ fast growth/ motile/ can mutate and transform/ eyespot detect light/ single chloroplast. Can switch between photosynthetic and heterotrophic growth |
| Chlorophyll light absorption | Solves thermodynamically unfavourable reaction Chl -> Chl* + A -> Chl+ + A- ->Chl Light absorbed at Chl -> Chl* + A |
| Charge separation | Chl* + A -> Chl+ + A- Only occurs if correct chemical environment Can extract e from H2O Key event converting light energy to chemical energy |
| Chlorophyll | A (methyl) and B(formyl) absorb different wavelength - red and blue Lipid soluble - position and orientation in membrane Neither alone provides sufficient energy Emerson Enhancement Effect -> work together to produce 200kjmol needed= |
| Z scheme | 2 inputs of energy (each photosystem) Only place of photochemical events All others thermodynamically unfavourable |
| Photosystem consists of light harvesting complex and reaction centre operating together | Electron transfer 10^5 x quicker than light capture only 2 of 36 do charge separation - "special pair" reaction centre surrounded by light harvesting chlorophylls- absorb photons and pass energy to reaction centre through RET fast and biophysical - no e move 99% efficiency Acceptor really close to special pair hence can grab to move to ETC |
| Reaction centre | dimer of reaction centre chlorophylls in luminal side Symmetry, but only 1 branch used Precise arrangement of electron acceptors facilitate electron transfer Light absorption in harvesting complexes in PII causes energy transfer to special pair in reaction centre by RET Charge separation and photochemistry due to carefully positioned electron acceptors |
| Electron acceptors in PII | Pheophytin and quinone |
| Photosynthetic electron transport chain | Electron transferred between internal electron carries in PSII reaction centre Quinone B accepts 2e and 2H+ from stroma, released from PSII as PQH2. lipid soluble PQH2 moves to cytochrome B6F complex - donates e and H+ move to lumen Electrons passed to plastocyanin - take e to PSI to replace loss by charge separation RET at PSI and charge reparation at reaction centre. Electron from P700 transferred to internal carriers inc. FeS clusters Electrons passed from terminal FeS cluster to stroll ferrodoxin |
| Role of Ferrodoxin | Ferrodoxin-NADP+ reductase catalyses transfer fo electrons from Fd to terminal electron acceptor, generating NADPH |
| 3 challenges of carbon fixation | Low atmospheric CO2 concentration CO2 fixation coordinated with light capture Enzymes carry out side reactions |
| Discovery of CBB cycle | Radiative tracer - C Placed algae in light then kill at intervals using boiling EtOH Analyse with paper chromatography to see what present 1 = triose phosphate, PEP, PGA 2= sucrose/ sugars 3= organic acids - malic/ citric 4= amino acids |
| RuBisCO | Ribulose Bisphosphate Carboxylase/ Oxidase Kcat = 3CO2/ sec Km = 10um CO2 (cell only 9um) 50% protein is RuBisCO as slow Carbon fixation by rubisco RuBP -> endiolate intermediate -> unstable intermediate -> 2 x 3-phosphoglycerate 8 large and 8 small subunits - active site in large subunit |
| The cycle | CO2 fixation by rubisco- no carriers Reduction of 3PGA to 3GAP - energy from ATP and NADPH needed RuBP regenerated using ATP Autocatalytic - self contained in use of carbon |
| Regulation of cycle | Cycle activated by light to ensure active when NADPH and ATP being made Some enzymes redox regulates (sedoheptulose-1,7-bisphosphate) Electrons from PSI reduce ferredoxin. Pass via thioredoxin to reduce inhibitory disulphide bridge to active enzyme Oxygen evolving complex -> manganese cluster buffers redox change. Only when 4 flashes of light and completely oxidised will water be split |
| Side reactions of RUBISCO | Oxygenation Misfire |
| Oxidation | Forms 13PGA and 1 phosphoglycollate instead of 2 3PGA Phosphoglycollate toxic hence rapidly metabolised by photorespiration Occurs in chloroplast, mitochondria and peroxisome - metabolically costly as ATP used and CO2 release din mitochondria. NADH used and H2O2 used - less carbon fixed and some wasted |
| Misfire reactions | XuBP generated from RuBP XuBP abhorrent product - damaged metabolite hence enters metabolite repair pathways XuBP inhibitor of RuBP |
| Repair Pathways | Rome aberrant product and return damaged molecule back to original e.g. NADPX from spontaneous chemical or enzyme reactions NADPH hydrate inhibits other enzymes, repair and remove R and S epimer in 2 part process ATP dependent NADPX dehydrates reconverts S epiterase, epimerise interconverts R and S epimers |
| Pre emption | Stop damage from unwanted metabolites occurring, rapid conversion of harmful to normal product XuBP inhibits RuBiSCO- harm due to accumulation prevented by phosphate to convert to Xu5BP- normal part of CBB cycle |
| Overview | Life based on ATP adn electron and H+ carriers ETC extract energy from biological molecules and store in ATP Photosystems rely on chlorophylls in LHC and RC doing different things Nothing of chemistry disappears in biology |
Want to create your own Flashcards for free with GoConqr? Learn more.
